Expi293 membrane pro crystal structure
Previous studies have hinted at TMEM16K being localised intracellularly (11) hampering investigation into any potential PLS or ion flux activity which traditionally rely on plasma membrane expression to easily characterise using conventional techniques. Images from Bodranghien et al (2017) (10) TMEM16Kĭespite having a wide expression profile, little is known about TMEM16K’s cellular role. 1 MRI Images showing cerebellar atrophy in (a) SCAR10 Patient and (b) healthy individual. Many patients also report a Co-enzyme Q10 deficiency (7).įig. 2) with resulting symptoms including cognitive impairment, deterioration of motor skills and epilepsy. TMEM16E mutation has been linked to disorders of the musculoskeletal system specifically gnathodiaphyseal dysplasia (4) and limb girdle muscular dystrophy (5,6). TMEM16K ( ANO10) mutation is linked to autosomal recessive spinocerebellar ataxia, type 10 (also known as SCAR10 or ARCA3) (7-9). SCAR10 patients display progressive atrophy of the cerebellum ( Fig. Mutation of TMEM16F causes Scott Syndrome (3), a blood coagulation disorder linked to reduced presentation of phosphatidylserine (PS) on the surface of platelet cells a key trigger for clot formation. Genetic disruption of TMEM16 PLS activity forms the basis of several inherited disorders. These grooves are lined with polar residues, providing a path for lipids to traverse the membrane through transient contacts with hydrophilic lipid headgroups. Structural investigation of TMEM16 homologues (2) identified a recessed hydrophilic ‘groove’ formed by several of TMEM16’s transmembrane helices as the main facilitator of its PLS activity. The TMEM16 family of membrane proteins were originally identified as a class of calcium activated chloride channels called anoctamins, with TMEM16A being the most well studied member of the family (1). Subsequently, many TMEM16 proteins were found to also act as phospholipid scramblases, which are sensitive to the concentration of calcium and have non-specific ion channel activity.
1 TMEM16-mediated Lipid Scrambling and non-specific ion channel activity
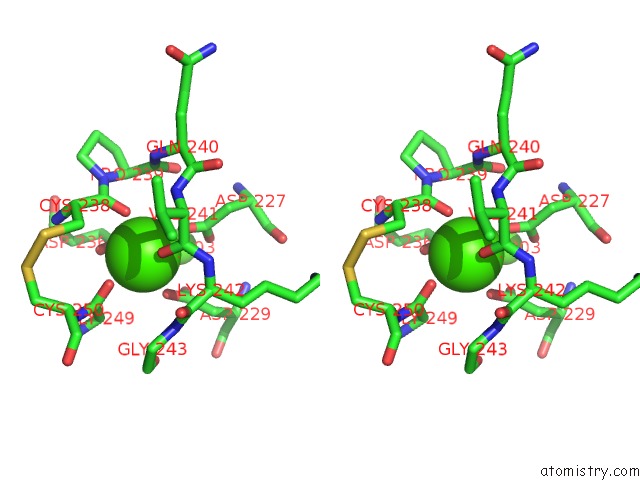
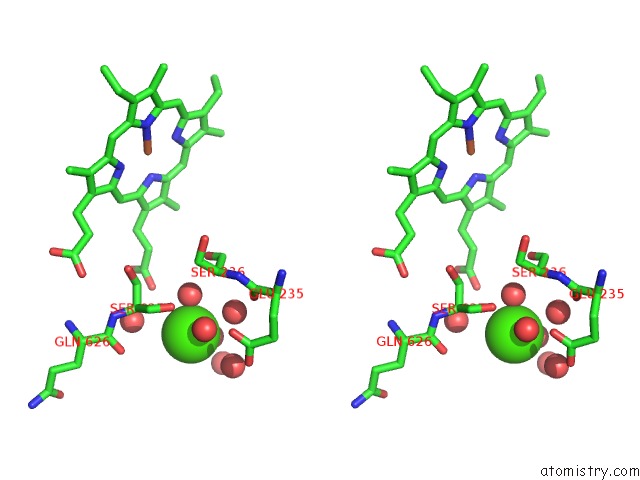
However, the nature of the phospholipid scramblases in the ER was not clear.įig. Many lipids are synthesised on the cytoplasmic face of the ER and yet the ER has a relatively even distribution of lipids. Phospholipid scramblase (PLS) proteins are integral membrane proteins that abolish lipid asymmetry by facilitating the two-way transfer of lipids between membrane envelopes along concentration gradients. Lipid bilayers surround cells and organelles and in most membranes, with the exception of the endoplasmic reticulum (ER), the composition of the two leaflets of the bilayer differ. Our highest resolution cryo-EM structure for TMEM16K allowed us to identify a bound lipid in the cavity behind the groove that transports the lipid headgroups and this lipid binding site may represent an allosteric modulator site, providing a direction for the design of binders which could modulate TMEM16K activity in cells. We also showed that TMEM16K mainly resides in the endoplasmic reticulum where it may be regulated by the ER’s unique lipid profile. We were able to show that TMEM16K acts as a lipid scramblase with non-selective ion channel activity that is sensitive to both Ca 2+ and lipid chain lengths. Through collaborations, we have investigated TMEM16K’s function and location in cells.
We have solved several structures of human TMEM16K through X-ray crystallography and cryo-EM capturing both active and inactive conformational states. SCAR10 is a late-onset neurodegenerative disorder which causes marked atrophy of the cerebellum with consequential deterioration in limb co-ordination, speech and eye movement. TMEM16K was a relatively understudied member of the family despite the observation that mutations in TMEM16K have been linked to the genetic disease autosomal recessive spinocerebellar ataxia Type 10 (Also known as SCAR10 or ARCA3). Although the first members of this family to be discovered, TMEM16A and TMEM16B, have a calcium-regulated chloride channel function, subsequently other members of the family, such as TMEM16F, were found to have lipid scramblase activity combined with non-selective ion channel activity. There are ten members of the TMEM16/Anoctamin family of proteins in mammals.
